 Oncolytics Biotech Inc. (NASDAQ: ONCY) just reasserted its position on the map again. The company has announced test results showing tumor shrinkage data from its U.S. Phase II clinical trial in patients with squamous cell carcinoma of the lung. This is a lung cancer study with its intravenous administration of REOLYSIN in combination with carboplatin and paclitaxel. If this translates anywhere close to this result in the later stages, then a feasible treatment for lung cancer may be closer to fruition.
Oncolytics Biotech Inc. (NASDAQ: ONCY) just reasserted its position on the map again. The company has announced test results showing tumor shrinkage data from its U.S. Phase II clinical trial in patients with squamous cell carcinoma of the lung. This is a lung cancer study with its intravenous administration of REOLYSIN in combination with carboplatin and paclitaxel. If this translates anywhere close to this result in the later stages, then a feasible treatment for lung cancer may be closer to fruition.
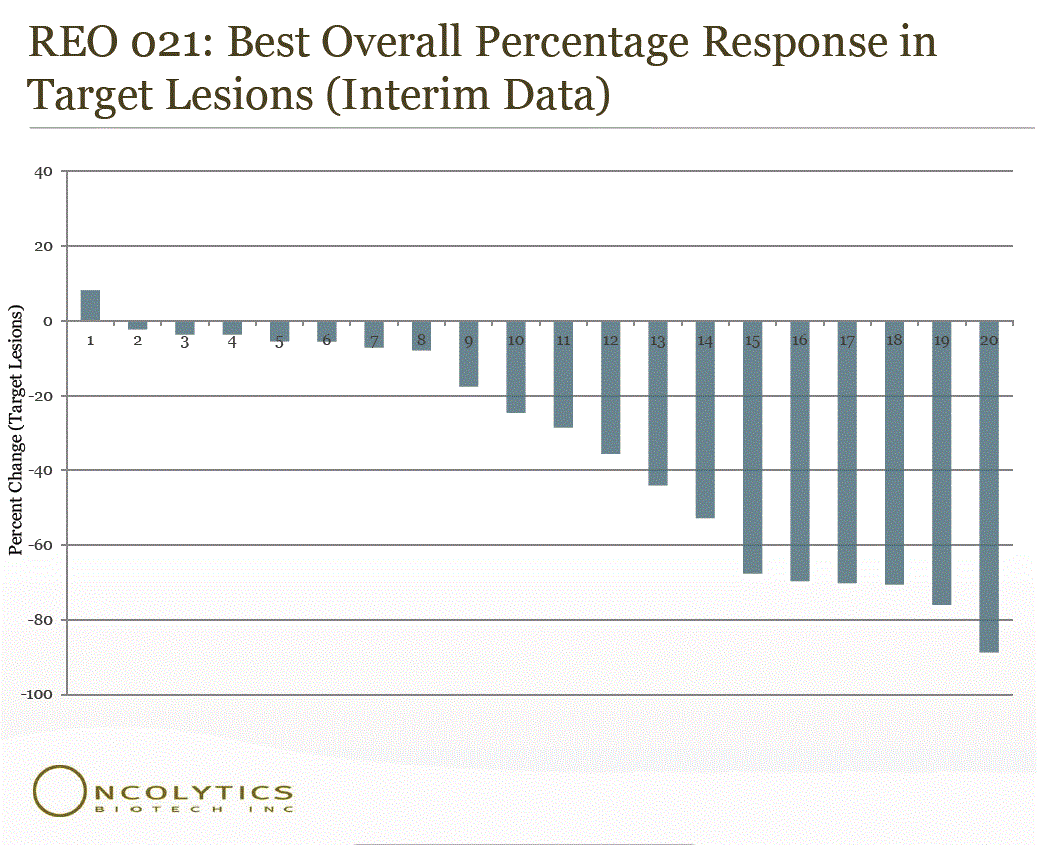
The company showed that 19 of 20 evaluable patients exhibited overall tumor shrinkage at 33.7% shrinkage. That is 95% of patients, but we would remind that this is only a 20 patient study, and that is far too small of a sample to make the case for a cure for lung cancer yet. That being said, this appears to be great news and the graph below should show some additional detail.
Outside of the obvious benefits, here is why this matters so much: Lung cancer is by far the leading cause of cancer death among both men and women and accounts for about 27% of all cancer deaths. More people die of lung cancer than from colon, breast and prostate cancers combined. Oncolytics used figures from the American Cancer Society in 2013 it is estimated that some 228,190 new cases of lung cancer will be diagnosed. Approximately 84% of all lung cancers are classified as non-small-cell lung cancer, and squamous cell carcinomas account for approximately 25% of all lung cancers. There will be an estimated 159,480 deaths from lung cancer in the United States in 2012.
Dr. Brad Thompson, President and CEO of Oncolytics said:
It’s exciting to have 95% of patients in this study exhibit tumour shrinkage and these results further suggest that REOLYSIN may have potential use in neoadjuvant (pre-surgical) settings. Based on these findings we intend to continue to look at REOLYSIN as a treatment for cancers of the lung and cancers that metastasize to the lung.
The primary endpoint of the study is objective tumor response rates, and the secondary objectives include progression free survival and overall survival. To date, the company has observed nine partial responses (PR), nine stable disease (SD) and three progressive disease (PD) by RECIST criteria for a disease control rate (complete response (CR) + PR + SD) of 86%. The study continues to enroll patients.
Before the effects of the premarket pop here, Oncolytics had a market cap of $273 million and investors should consider that Oncolytics is based in Calgary, Canada. Shares are indicated up 33% at $4.76 against a 52-week range of $1.60 to $5.29.