
The 2010s will go down in history as a decade of many newsworthy health-related stories, many of which were not good news — Ebola, measles, antibiotic resistance. But in the years since 2010 there were also many promising discoveries in medicine, life-saving drugs approved, and great strides taken addressing national health crises. Some of these stories will have lasting effects for generations to come.
24/7 Tempo reviewed multiple news archives and dozens of articles published since 2010 to select 15 of the most positive health news stories that made headlines.
Some of the most talked about stories over the last few years have influenced health guidelines, treatment of serious disease, and even government policy.
Reports of significant research developments in the treatment and prevention of chronic and other conditions gave hope to millions of Americans. Some of the good news broke as recently as just a few months ago – these are the 15 biggest health topics of 2019.
Click here for 15 of the best health news over the last decade.
CT scans in high risk patients can reduce overall lung cancer mortality
> Year: 2011
> Category: Diseases
The tremendous effort by researchers and health institutions to develop a cure for cancer over the decades since the legislation for the War on Cancer was enacted in 1971 will likely continue. Any good news on developments are worth noting. The 2011 National Lung Screening Trial showed a reduction in lung cancer mortality of 20% in high risk patients receiving low-dose CT (LDCT) compared to chest X-ray. The CDC recommends that people at high risk of developing lung cancer — heavy smokers, people who have smoked as recently as 15 years, and people who are 55 years or older — undergo annual LDCT scans because of potential risks..
In 2016, there were 218,229 new cases of lung cancer, and 148,869 people died from the disease in the United States, according to the CDC. The American Cancer Society estimates 142,670 deaths from lung cancer in 2019. A major reason for the disease’s high mortality rate is that the tumor does not typically cause symptoms until it spreads, making early screening especially crucial to improving survival rates.
[in-text-ad]
Melanoma drug approved
> Year: 2011
> Category: Treatment
After more than a decade of no new potential drugs for melanoma, the deadliest form of skin cancer, the FDA approved vemurafenib, sold under the brand name Zelboraf, in 2011 for patients with metastatic melanoma with the BRAF(V600E) mutation or for those who have tumors that cannot be surgically removed.
Zelboraf was seen as a major development because it can improve melanoma patients’ quality of life — the drug is a simple pill taken twice a day — and it may extend survival rate. In a trial, the length of time melanoma patients who received Zelboraf lived without the cancer getting worse was almost double the length of patients who did not take the drug.
Since 2011 several drugs have been approved to treat melanoma, and survival rates of this deadly cancer have improved.

Gene editing is now possible
> Year: 2012
> Category: Technology
Gene editing is the process of changing an organism’s DNA. After decades of research around the world, scientists made a major breakthrough with the discovery of clustered repeats of DNA sequences, known as CRISPR.
First described in 2012, CRISPR, or Clustered Regularly Interspaced Short Palindromic Repeats, is the basis for potentially world-changing gene editing technology, or, as some might say, DNA hacking. It may be used to develop treatments for a range of diseases, including cancer and genetic disorders. In 2015, CRISPR was successfully used for the first time to save a life. Two baby girls, 11-month and a 16-month-old, received gene editing treatment to help them fight leukemia.
While the the gene-altering tool is bringing revolutionary change to health fields, it has also raised serious ethical concerns. Misuses and inadvertently harmful uses of CRISPR include those for creating designer babies, and causing environmental ripple effects by eliminating disease-spreading insects.

FDA says trans fat should not be considered ‘safe’
> Year: 2013
> Category: Eating
Trans fats, or partially hydrogenated oils, have been widely used for years, most notably in fast foods. Trans fats can raise the levels of “bad” LDL cholesterol, lower the levels of good-for-you HDL cholesterol, and increase the risk of heart disease, the No. 1 killer in the United States. And now they are on their way out.
In 2013, the FDA officially announced trans fats should not be considered safe in human food. In 2015, the agency gave food manufacturers three years to phase out the use of trans fats in their products. The deadline was June 18, 2018, although the FDA granted a one-year extension in the use of artificial trans fats in some cases. The ban will be fully implemented in Jan. 1, 2020.
[in-text-ad-2]

HIV prevention pill
> Year: 2014
> Category: Diseases
About 50,000 Americans are diagnosed with HIV every year, according to the CDC. Despite advancements in treatment and years of research into the infection, HIV does not have a cure. In 2014, the CDC issued new guidelines that recommend a pill to people at high risk of HIV as a prevention method. High risk people include gay or bisexual men, injection drug users, and women with an HIV+ partner.
The agency said that the pill, sold under the brand name Truvada, may lower the risk by as much as 90% when taken consistently. Truvada has been used to treat HIV since 2012 when the FDA approved the drug. Truvada contains tenofovir and emtricitabine, which when used in combination with other antiviral medication may keep the HIV virus from establishing a permanent infection.
A new way to treat cavities
> Year: 2015
> Category: Treatment
In 2015, the FDA approved a painless new way to treat tooth decay called silver diamine fluoride (SDF). It’s a liquid that is applied directly to cavities to stop the decay. The FDA gave it a “breakthrough therapy designation” two years later.
As a non-invasive and fairly cheap method (it costs about $20-$25 per tooth), SDF treatment, which must be prescribed by a dentist, can save people a lot of money. About 91% of American adults have dental decay, and about 27% have untreated tooth decay, according to the CDC. Tooth decay is common among kids as well — it’s the most common chronic disease in children between 6 and 11 years of age.
[in-text-ad]

3D printing of human organs
> Year: 2015
> Category: Technology
3D printing technology has improved considerably over the past few years. (Today, low-budget 3D printers are available for anyone who can spare $100.) The technology has advanced so much that producing fully functional replacement organs from a person’s own cells seems like a not so distant possibility. Scientists at Harvard’s Wyss Institute have grown a heart tissue that beats just like a normal human heart.
Production for treatment is still years away, however. The technique, called sacrificial writing into functional tissue (SWIFT), has not even been tested on mice yet. But if it works, it can be used to print other organs, too, potentially saving the lives of thousands of people who are waiting for an organ transplant.
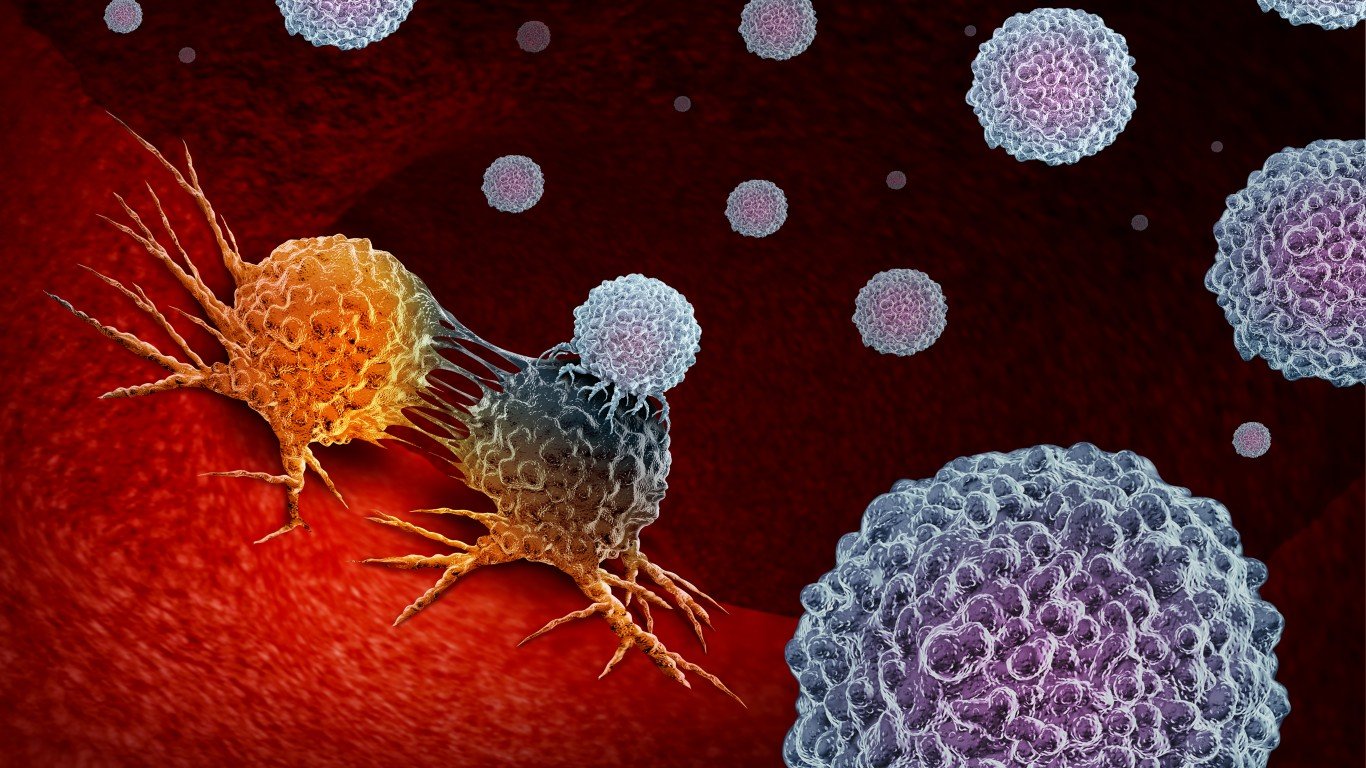
Immunotherapy and cancer
> Year: 2016
> Category: Treatment
Cancer immunotherapy was named the 2016 Advance of the Year by the American Society of Clinical Oncology. The therapy is designed to support and boost the immune system’s response to cancer cells, rather than targeting the cancer itself. One of the most successful immunotherapies so far is the checkpoint inhibition. It makes the immune response stronger by keeping immune cells activated, which does not normally happen when a person has cancer.
It may take decades until immunotherapy could replace the current standards in cancer treatment of surgery, chemotherapy, and radiation, but currently hundreds of immunotherapy drugs are being tested in clinical trials on people.
Some benefits of immunotherapy include fewer side effects than radiation or chemotherapy, lower risk of relapse, and making other cancer treatments more effective.

Opioid crisis recognized as national public health emergency
> Year: 2017
> Category: Public health
Every day over 130 people in the United States die from opioid overdose, including pain medication, heroin, and synthetic opioids such as fentanyl, according to the National Institutes of Health. In 2017, President Donald Trump declared the opioid crisis a national public health emergency, giving hope that the federal government’s involvement could help fight the worst drug crisis in U.S. history.
The official designation removed certain administrative requirements for accessing federal funds to fight the epidemic, including the use of taxpayers’ money to make addiction treatments and naloxone, a life-saving medication that can reverse an opioid overdose, drug, more accessible.
The Department of Health and Human Services has renewed the opioid crisis’ status as a national emergency several times since 2017. Money has been used to speed up a survey on whether and how often doctors prescribe opioids and help launch anti-addiction programs quicker, according to the a 2018 report by the Government Accountability Office.
[in-text-ad-2]

Early-stage Alzheimer’s treatment
> Year: 2019
> Category: Diseases
Currently, there is no treatment for Alzheimer’s disease, the sixth leading cause of death in the United States. Pharmaceutical companies and universities have tried to tackle different aspects of the neurodegenerative disorder, but to no avail. Until just a few months ago.
Biogen, a biotechnology company, announced in October 2019 it would ask the FDA to approve its Aducanumab drug as first treatment for early Alzheimer’s disease. The company said that patients in the early stages of the disease who were treated with a high dose of the drug experienced significant improvements in memory, orientation, and language. If Aducanumab is approved, it will be one of a handful of drugs approved to treat the disease.

Smoking rates at all-time low
> Year: 2018
> Category: Habits
The short and long-term health problems smoking causes have been well-documented for decades. Today cigarette smoking among U.S. adults is at an all-time low — 13.7% in 2018, according to the CDC.
While smoking regular cigarettes is down, smoking e-cigarettes is on the rise. About 37% of 12th graders reported vaping in 2018, compared with 28% in 2017. A recent Gallup survey found that 20% of 18- to 29-year-olds vape regularly, more than twice the national average for all age groups.
There has been a recent outbreak of lung injury associated with the use of e-cigarette. At least 47 deaths and 2,290 lung injuries have been confirmed by the CDC as a result of vaping as of Nov. 20, 2019. The agency has identified vitamin E acetate, an additive in some THC-containing e-cigarettes, as the likely cause for the lung injuries.
[in-text-ad]

Cystic fibrosis treatment approved by FDA
> Year: 2019
> Category: Treatment
About 30,000 Americans live with cystic fibrosis, a fairly common genetic disease that affects the lungs and other organs, limiting one’s ability to breathe as the disease progresses. About 1,000 new cases are diagnosed every year.
The FDA approved in 2019 what it called a “new breakthrough” therapy to treat the condition. The medication, sold under the name Trikafta, is available to patients who are 12 years or older and have the F508del mutation, the most common cystic fibrosis mutation. It is found in 90% of the people living with the disease. The treatment can increase the life expectancy of patients, which is now around 44 years.
Second HIV patient goes into remission
> Year: 2019
> Category: Diseases
A second person since HIV was identified in the 1980s has been said to be in sustained remission. The patient, who was treated in London, has not been given antiretroviral therapy for 18 months, and the virus has remained undetectable. The good news comes more than a decade after the “Berlin patient,” known as the first person to have been “cured” from the infection. Both patients received a stem cell transplant.
HIV, the virus that causes AIDS, is one of the most serious global health challenges. Almost 38 million people live with HIV worldwide, according to the World Health Organization. Just over 60% are receiving treatment.

Blood test detects breast cancer 5 years early
> Year: 2019
> Category: Diagnoses
Even though deaths from breast cancer have declined, the disease remains the second leading cause of cancer death among women in the United States, according to the CDC. More than 40,000 women die from it a year.
Improved rates of early detection have helped drive up survival rates. A recent British study offers hope that the condition could now be detected five years before there are any clinical signs of it. The new method is a blood test that identifies the body’s immune response to antigens produced by tumor cells. The test may be available in clinics in about five years.
[in-text-ad-2]
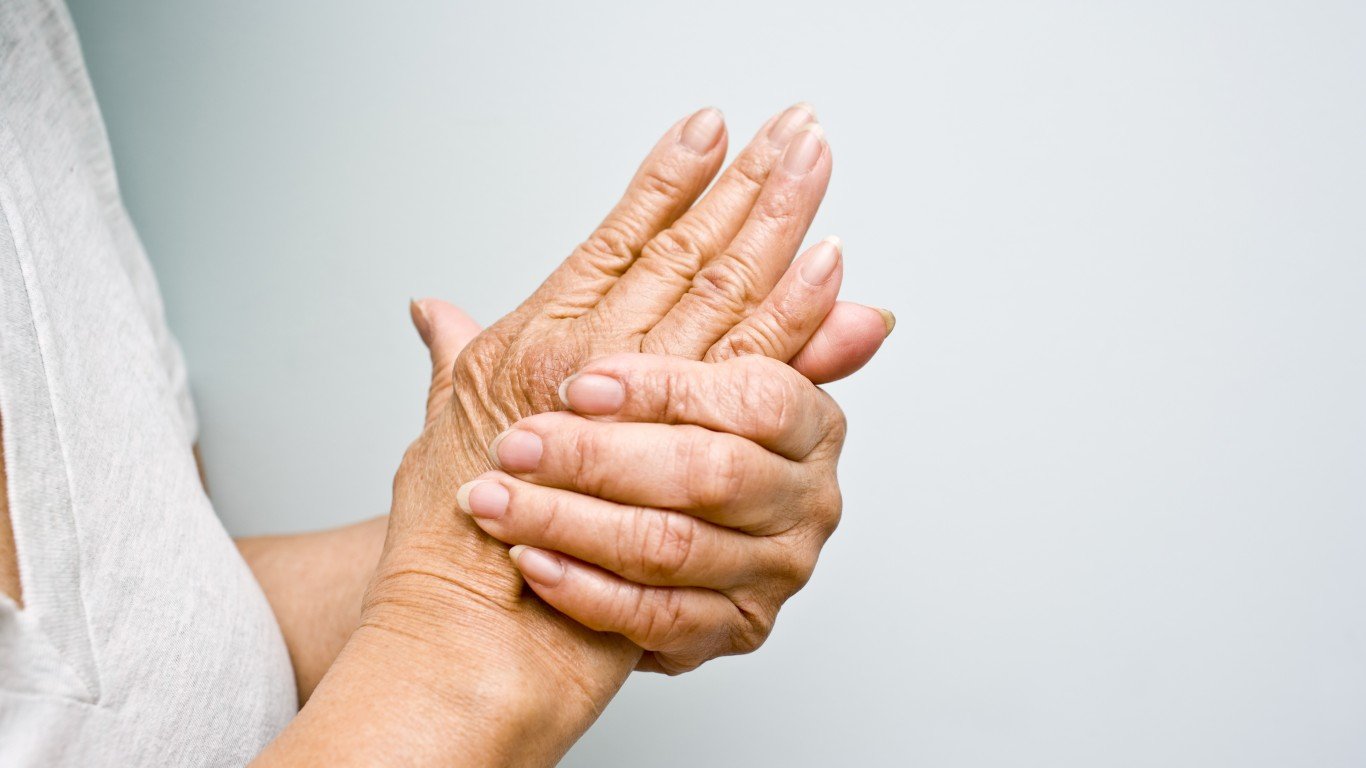
Finding a cure for arthritis
> Year: 2019
> Category: Treatment
2019 has been an exciting year in the field of health technology and scientific research. In addition to such technological developments as organ printing and gene editing, recent research has shown promise for a cure for arthritis. Millions of people suffering from joint inflammation — from osteoarthritis, for example, which is the most common form of arthritis — may be helped.
A recent study published in the Science Advances journal has found that “cartilage in human joints can repair itself […] to regenerate limbs.” The body was previously believed to be unable to do so. People have a molecule that helps with joint tissue repair, and that molecule is more active in ankles and less active in knees and hips. The findings can help develop treatments that may prevent, slow, or even reverse arthritis.
Sponsored: Want to Retire Early? Here’s a Great First Step
Want retirement to come a few years earlier than you’d planned? Or are you ready to retire now, but want an extra set of eyes on your finances?
Now you can speak with up to 3 financial experts in your area for FREE. By simply clicking here you can begin to match with financial professionals who can help you build your plan to retire early. And the best part? The first conversation with them is free.
Click here to match with up to 3 financial pros who would be excited to help you make financial decisions.
Thank you for reading! Have some feedback for us?
Contact the 24/7 Wall St. editorial team.
 24/7 Wall St.
24/7 Wall St.