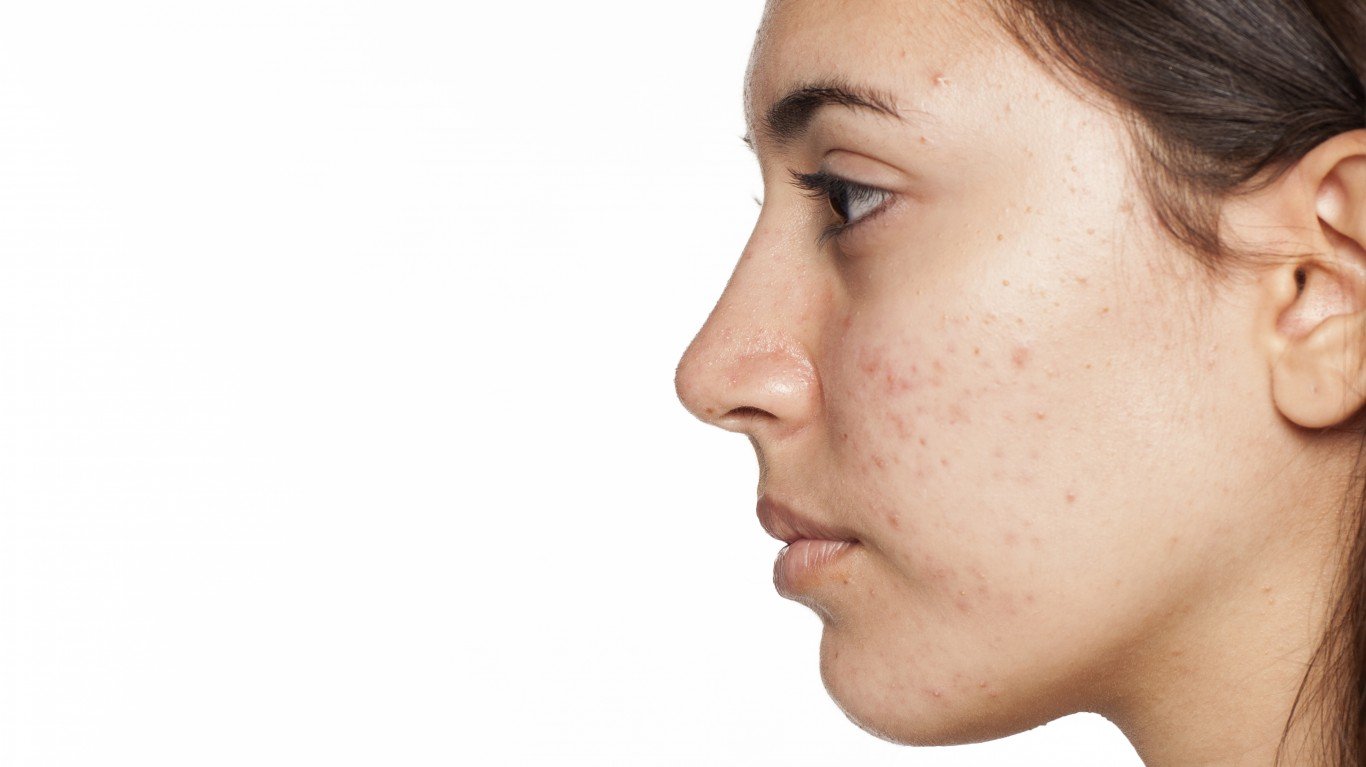
Pfizer Inc. (NYSE: PFE | PFE Price Prediction) has announced positive top-line results from a second Phase 3 pivotal study evaluating abrocitinib in patients aged 12 and older with moderate to severe atopic dermatitis (AD).
This is the second monotherapy trial in the Janus kinase 1 (JAK1) Atopic Dermatitis Efficacy and Safety (JADE) global development program (B7451013, or JADE MONO-2). Pfizer announced positive top-line results from the first trial in the JADE program (B7451012, or JADE MONO-1) back in May 2019.
Consistent with JADE MONO-1, results showed that by week 12 the percentage of patients achieving each co-primary efficacy endpoint and each key secondary endpoint with either dose of abrocitinib was statistically significantly higher than placebo.
Additionally, a statistically significant number of patients achieved a reduction in pruritus by week two, as measured by a four-point or larger reduction in itch severity measured with the pruritus numerical rating scale (NRS).
Michael Corbo, Ph.D., Chief Development Officer, Inflammation & Immunology, Pfizer Global Product Development, commented:
These findings add to a growing body of evidence supporting the potential of abrocitinib to improve the lives of people living with moderate to severe atopic dermatitis. We look forward to continued findings from the JADE program, with results from the next abrocitinib efficacy study, using an active control, becoming available in spring 2020. This will further our understanding of abrocitinib as a potential medicine for patients who suffer from this chronic condition.
[nativounit]
Shares of Pfizer traded up fractionally early Friday at $36.09, in a 52-week range of $33.97 to $46.47. The consensus analyst price target is $41.82.
[recirclink id=580615]
[wallst_email_signup]