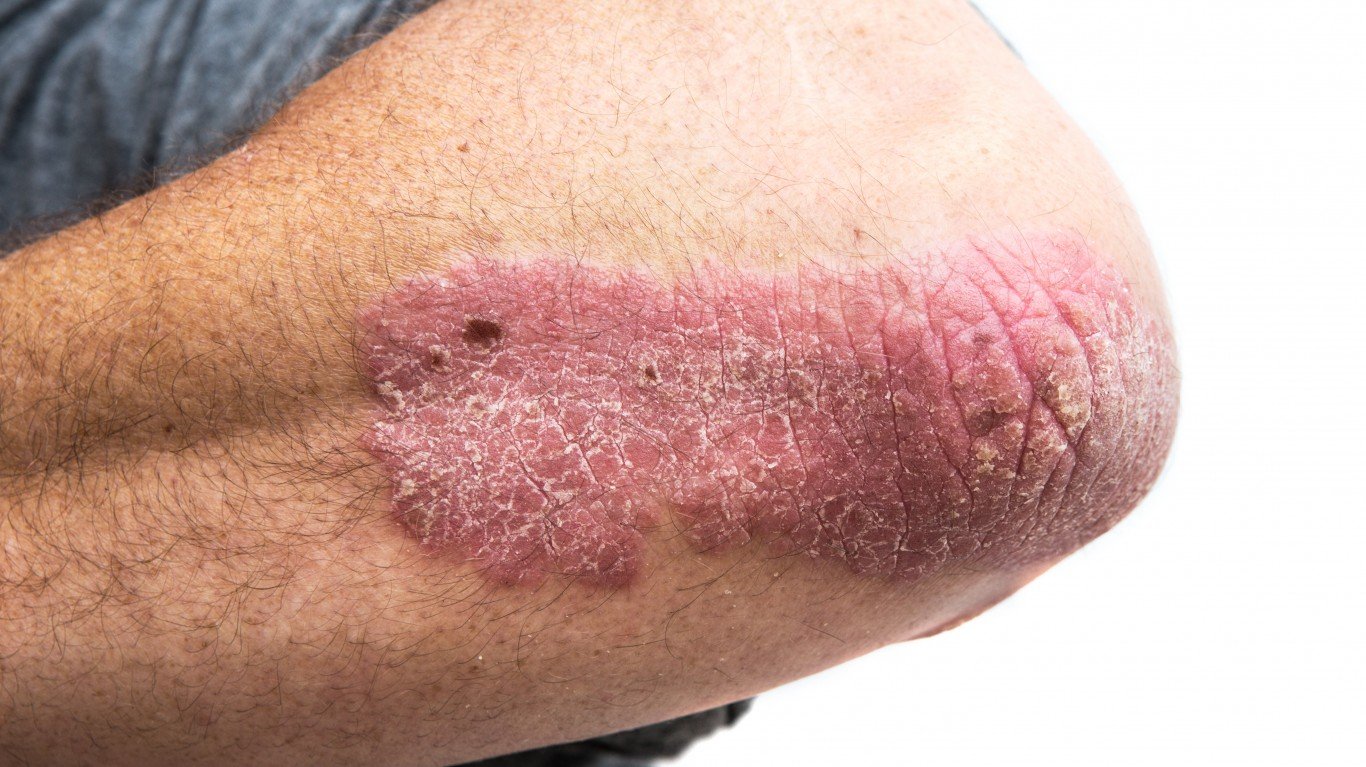
Eli Lilly and Co. (NYSE: LLY | LLY Price Prediction) announced some good news for people suffering moderate to severe plaque psoriasis when it presented at the Fifth Annual Maui Derm NP+PA Fall meeting. A total of 1,027 patients with moderate to severe plaque psoriasis were enrolled in the study to evaluate the efficacy and safety of Taltz compared to Tremfya.
Specifically, this data comes from Eli Lilly’s Phase 4 study, which was the first head-to-head study between an IL-17A inhibitor and an IL-23/p19 inhibitor using the Psoriasis Area Severity Index (PASI) as the primary endpoint.
Taltz met the primary endpoint of superiority over Tremfya in the proportion of patients with moderate to severe plaque psoriasis achieving complete skin clearance as measured by PASI 100 at Week 12 (41.3% versus 24.9%).
Additionally, all major secondary endpoints up to Week 12 were achieved. Key secondary endpoints included superiority over Tremfya in the proportion of patients achieving PASI 75 at Week 2, PASI 90 at Weeks 4 and 8, PASI 100 at Weeks 4, 8 and 24.
Taltz is approved for the treatment of adults with moderate to severe plaque psoriasis who are candidates for systemic therapy or phototherapy. Taltz is also approved for the treatment of adults with active psoriatic arthritis and active ankylosing spondylitis.
[nativounit]
Andrew Blauvelt, lead study investigator, M.D., M.B.A., dermatologist and president of Oregon Medical Research Center, commented:
Healthcare providers and patients value speed of response when evaluating treatment options for moderate to severe plaque psoriasis. The results from the IXORA-R study demonstrate that Taltz was effective in helping more patients achieve completely clear skin by Week 12, with a 50 percent improvement in skin plaques seen as early as Week 1.
Shares of Eli Lilly traded at $107.00, in a 52-week range of $104.17 to $132.13. The consensus price target is $124.25.
[recirclink id=582221]
[wallst_email_signup]