Ardelyx Inc. (NASDAQ: ARDX) shares dipped on Tuesday after the firm posted a win with the U.S. Food and Drug Administration (FDA). Overall, this seems to be a case of “sell the news” for Ardelyx.
When it comes down to it, companies winning approval from the FDA or beating a trial are almost expected in some cases. If investors already have priced this news into the stock, when it actually comes around they tend to sell the news, even if it’s good news. This is why ostensibly good news for companies like Ardelyx can yield a dip in the price.
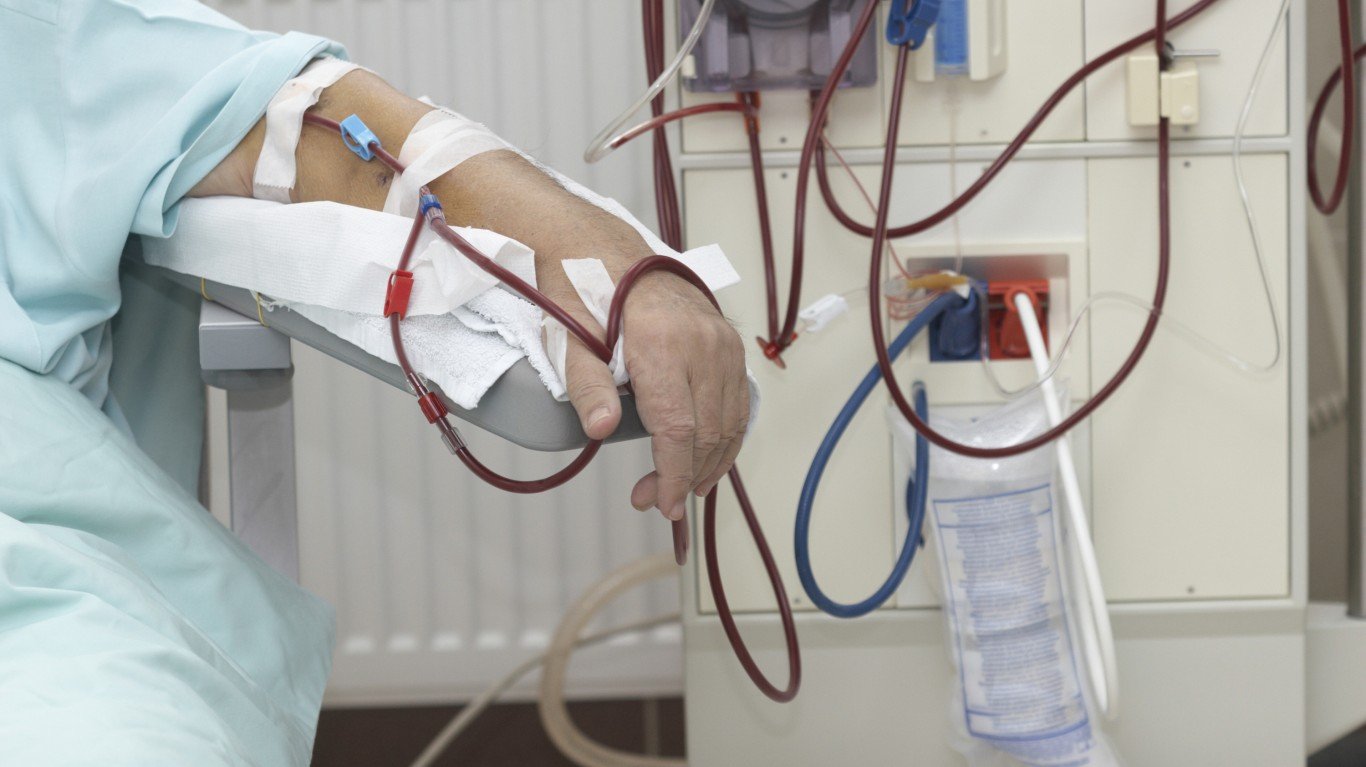
The announcement was that the FDA has accepted Ardelyx’s New Drug Application (NDA) of tenapanor for the control of serum phosphorus in adult patients with chronic kidney disease on dialysis.
The NDA is supported by three successful Phase 3 trials involving over 1,000 patients that evaluated the use of tenapanor.
[nativounit]
Management noted that the acceptance of this NDA is extremely exciting as it represents the next critical step toward bringing to market a completely new approach to the management of hyperphosphatemia, an area where a significant unmet need exists.
Looking ahead, the FDA has set a Prescription Drug User Fee Act (PDUFA) goal date of April 29, 2021.
With potential approval in the second quarter of 2021, the firm is continuing to advance commercial preparations for the launch of tenapanor. Again, management believes that with tenapanor, the firm has discovered and developed a therapy that will truly advance care for patients on dialysis.
Excluding Tuesday’s move, Ardelyx stock had underperformed the broad markets, retreating about 17% year to date. In the past 52 weeks, the share price actually was about 18% higher.
Ardelyx stock traded about 6% to $5.87 on Tuesday, in a 52-week range of $4.16 to $8.81. The consensus price target is $13.00.
[recirclink id=737721][wallst_email_signup]